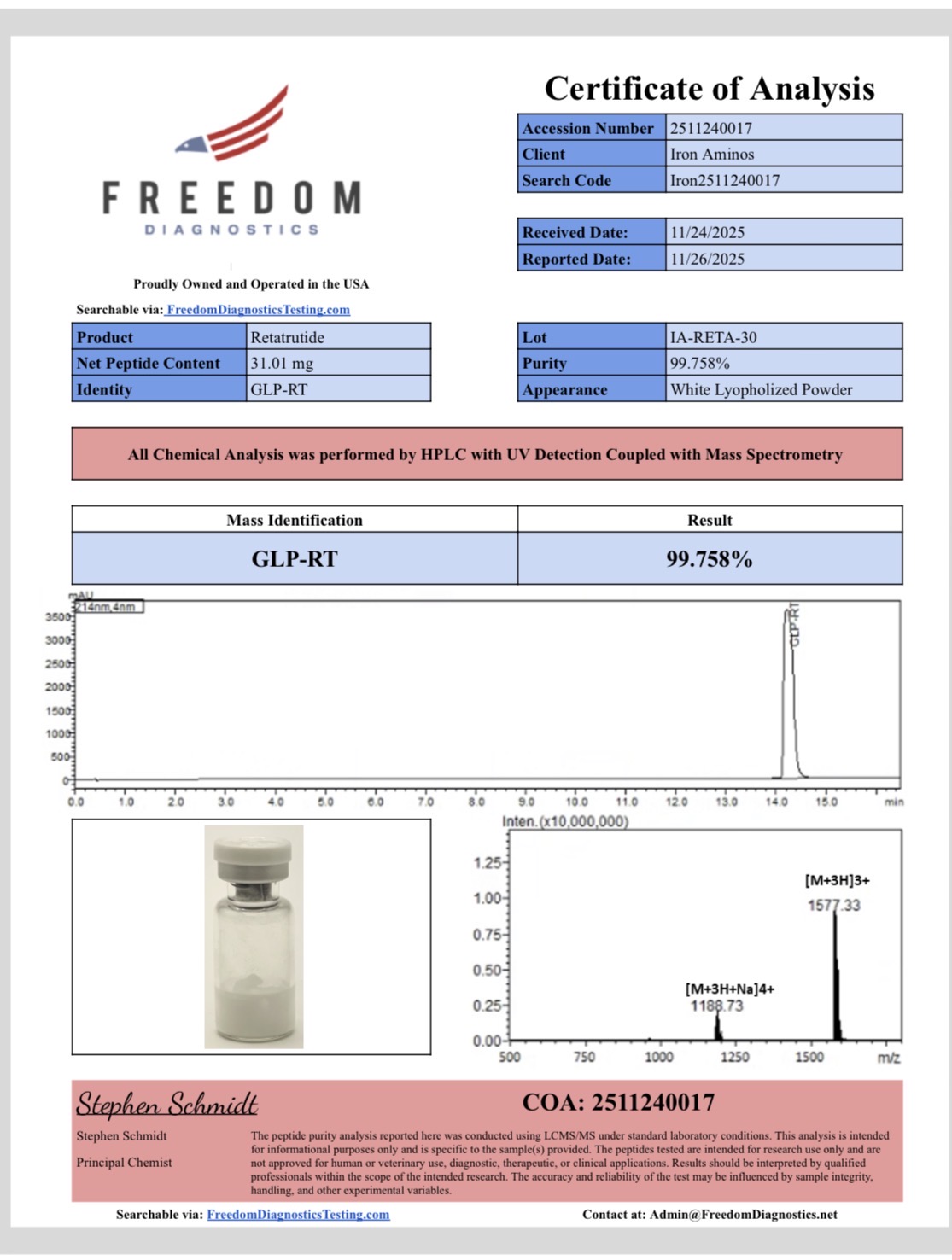
IRON AMINOS GLP-3 RETA 30MG
Research-grade incretin-pathway material prepared for consistency and clear documentation in professional lab workflows.
Key Features
-
30mg GLP-3 RETA per vial (lyophilized powder)
-
≥99% purity for reproducible results
-
Sterile, sealed glass vial with tamper-evident flip cap
-
Lot and batch labeling for traceability
-
Signature Iron Aminos red honeycomb label for quick identification
-
Diluent not included
Specifications
-
Compound: GLP-3 RETA
-
Class: Triple receptor agonist research category (GLP-1, GIP, glucagon)
-
Form and appearance: Lyophilized powder, white to off-white
-
Amount: 30mg per vial
-
Purity: ≥99%
-
Storage, lyophilized: Cool, dry, away from light
-
Storage, after reconstitution: Refrigerate and use promptly per internal SOP
What’s Included
-
1 × GLP-3 RETA 30mg vial (no diluent)
Handling
Use an appropriate sterile diluent and aseptic technique. Follow your lab SOPs for reconstitution, aliquoting, labeling, and disposal.
Research Snapshot
-
Triple agonists in this class have demonstrated large body-weight reductions versus placebo and active comparators in randomized studies using structured dose escalation and dietary guidance.
-
In type 2 diabetes research settings, this class has shown meaningful HbA1c reductions along with improvements in select metabolic markers.
-
Gastrointestinal adverse events such as nausea, vomiting, and diarrhea are commonly reported and tend to be dose related. Protocols frequently employ gradual titration to improve tolerability.
-
GLP-3 RETA is investigational and not approved for medical use; findings are model- and protocol-dependent.
Compliance
For research use only. Not for human consumption.
Not a drug, food, or cosmetic and not intended to diagnose, treat, cure, or prevent any disease.